metabolism hl
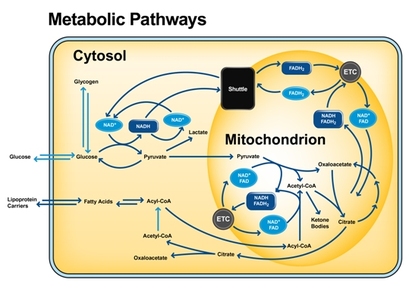
8.1.1 DEFINITION OF METABOLISM: Metabolic pathways consist of chains and cycles of enzyme-catalyzed reactions.
Note: By the break of the food (bonds), through metabolism we get energy ATP.
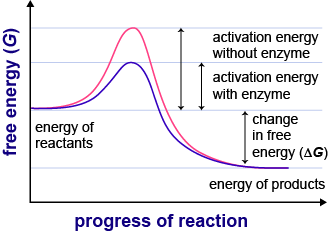
8.1.2 Enzymes lower the activation energy of the chemical reactions that they catalyze.
Note: By the break of the food (bonds), through metabolism we get energy ATP.
8.1.2 Enzymes lower the activation energy of the chemical reactions that they catalyze.
ACTIVATION ENERGY
-Is the amount of energy that must be put into a reaction to make it occur.
-An enzyme stresses the bonds in the substrate, reducing the activation energy required for a reaction to occur.
-Enzymes lower the activation energy of a reaction
-An enzyme stresses the bonds in the substrate, reducing the activation energy required for a reaction to occur.
-Enzymes lower the activation energy of a reaction
8.1.3 Enzyme inhibitors can be competitive or non-competitive.
Enzyme inhibitors are the only things that can stop an enzyme's reaction.
8.1.4 Metabolic pathways can be controlled by end-product inhibition.
Enzyme inhibitors are the only things that can stop an enzyme's reaction.
8.1.4 Metabolic pathways can be controlled by end-product inhibition.
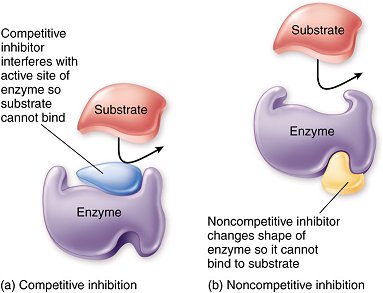
competitive inhibitors
•Substrate and inhibitor are chemically very similar
•Inhibitor binds to the active site of the enzyme, prevents substrate from binding
•Example: Folic acid synthetase
–Folic acid synthetase is an enzyme in bacteria which normally produces folic acid (vitamin) from PABA
–Sulfanilamides (antibiotics) occupies the active site of folic acid synthetase; blocks access of PABA
–Bacteria die without folic acid
•Inhibitor binds to the active site of the enzyme, prevents substrate from binding
•Example: Folic acid synthetase
–Folic acid synthetase is an enzyme in bacteria which normally produces folic acid (vitamin) from PABA
–Sulfanilamides (antibiotics) occupies the active site of folic acid synthetase; blocks access of PABA
–Bacteria die without folic acid
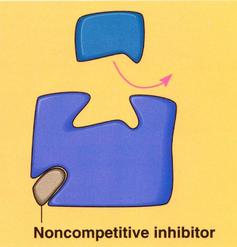
non-competitive inhibitors
•Substrate and inhibitor are not similar
•Inhibitor binds to the enzyme at different site than the active site
•The inhibitor changes the rate of the enzyme, causing slow enzyme activity
•Example: silver, Ag+
–silver forms bonds with the -SH groups of cysteine, the amino acid which forms covalent disulfide bridges
–the disruption of disulfide bridges alters the tertiary structure of the enzyme, affecting its active site
•Inhibitor binds to the enzyme at different site than the active site
•The inhibitor changes the rate of the enzyme, causing slow enzyme activity
•Example: silver, Ag+
–silver forms bonds with the -SH groups of cysteine, the amino acid which forms covalent disulfide bridges
–the disruption of disulfide bridges alters the tertiary structure of the enzyme, affecting its active site
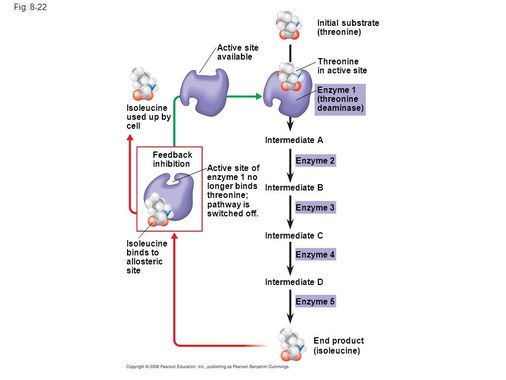
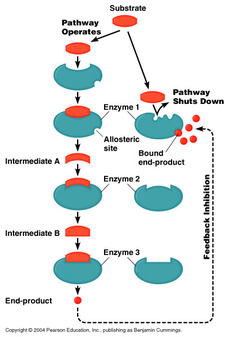
end product inhibition
•When the end product is formed in excess, the excess products interact with enzymes at the beginning of the pathway à decrease enzyme activity
•Negative feedback = the rate of the process decreases as the concentration of the product increases.
•Example: Threonine to isoleucine (amino acids)
NOTE: this inhibitor is used when there is an excess of the final products in the body, then this inhibitor joins the enzyme for safety and stops or slows the enzyme activity. If there is a lack of this inhibitor can causes diseases like diabetes or faints.
The own product of the reaction acts as the inhibitor.
•Negative feedback = the rate of the process decreases as the concentration of the product increases.
•Example: Threonine to isoleucine (amino acids)
NOTE: this inhibitor is used when there is an excess of the final products in the body, then this inhibitor joins the enzyme for safety and stops or slows the enzyme activity. If there is a lack of this inhibitor can causes diseases like diabetes or faints.
The own product of the reaction acts as the inhibitor.
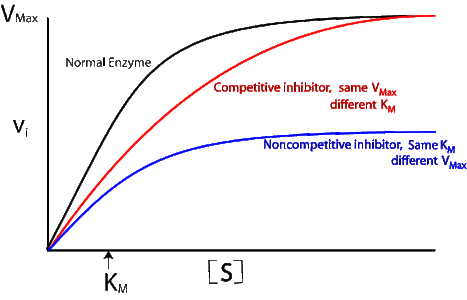
distinguish between the different types of inhibition
•Competitive inhibition: more substrate, greater rate of reaction
•Non competitive inhibition: prevents enzyme binding regardless of substrate concentration
•V = rate of reaction
•[S] = substrate concentration
•Non competitive inhibition: prevents enzyme binding regardless of substrate concentration
•V = rate of reaction
•[S] = substrate concentration
Theory of Knowledge: Many metabolic pathways have been described following a series of carefully controlled and repeated experiments. To what degree can looking at component parts give us knowledge of the whole?
Looking at the parts of the components is big helpful toll, if we spent enough time looking at them we will be able to memorize them, comprehend the structure and gain new knowledge. I believe that it is really important to spent some time looking at the components until we understand why it is that that specific part is there and what does it do to the whole component..
Guidance: Enzyme inhibition should be studies using one specific example for competitive and non-competitive inhibition.
All the information from: Power point and Pearson's book.
Looking at the parts of the components is big helpful toll, if we spent enough time looking at them we will be able to memorize them, comprehend the structure and gain new knowledge. I believe that it is really important to spent some time looking at the components until we understand why it is that that specific part is there and what does it do to the whole component..
Guidance: Enzyme inhibition should be studies using one specific example for competitive and non-competitive inhibition.
All the information from: Power point and Pearson's book.
| 8.1_metabolism_1.pptx | |
| File Size: | 3957 kb |
| File Type: | pptx |