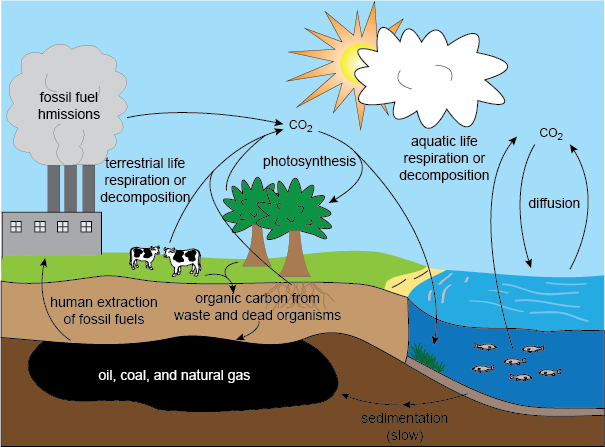
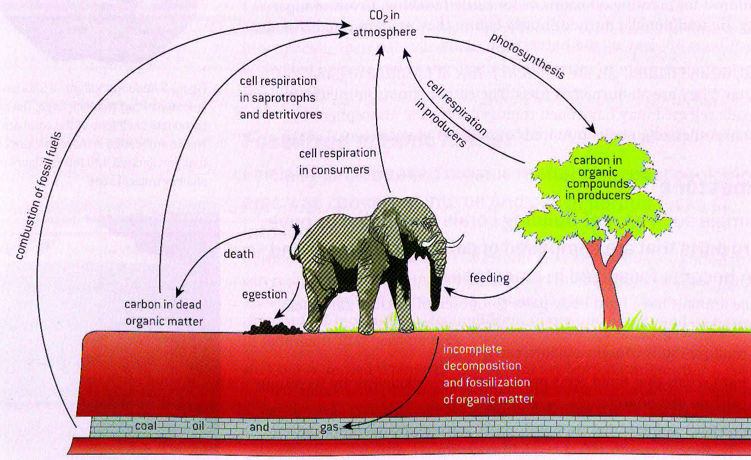
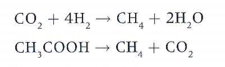CARBON CYCLING
Source: Pearson and Oxford text. Carbon cycling
4.3.1 Autotrophs convert carbon dioxide into carbohydrates and other carbon compounds
4.3.2 In aquatic ecosystems, carbon is present as dissolved carbon dioxide and hydrogen carbonate ions
4.3.3 Carbon dioxide diffuses from the atmosphere or water into autotrophs
4.3.4 Carbon dioxide is produced by respiration and diffuses out of organisms into water or the atmosphere
4.3.5 Methane is produced from organic matter in anaerobic conditions by methanogenic archaeans and some diffuse into the atmosphere or accumulates in the ground.
4.3.6 Peat forms when organic matter is not fully decomposed because of acidic and/or anaerobic conditions in waterlogged soils.
4.3.7 Partially decomposed organic matter from past geological eras was converted either into coal or into oil and gas that accumulates in porous rocks.
4.3.8 Carbon dioxide is produced by the combustion of biomass and fossilized organic matter
4.3.9 Animals such as reef-building corals and Mollusca have hard parts that are composed of calcium carbonate and can become fossilized in limestone.
4.3.1 Autotrophs convert carbon dioxide into carbohydrates and other carbon compounds
4.3.2 In aquatic ecosystems, carbon is present as dissolved carbon dioxide and hydrogen carbonate ions
4.3.3 Carbon dioxide diffuses from the atmosphere or water into autotrophs
4.3.4 Carbon dioxide is produced by respiration and diffuses out of organisms into water or the atmosphere
4.3.5 Methane is produced from organic matter in anaerobic conditions by methanogenic archaeans and some diffuse into the atmosphere or accumulates in the ground.
4.3.6 Peat forms when organic matter is not fully decomposed because of acidic and/or anaerobic conditions in waterlogged soils.
4.3.7 Partially decomposed organic matter from past geological eras was converted either into coal or into oil and gas that accumulates in porous rocks.
4.3.8 Carbon dioxide is produced by the combustion of biomass and fossilized organic matter
4.3.9 Animals such as reef-building corals and Mollusca have hard parts that are composed of calcium carbonate and can become fossilized in limestone.
carbon
4.3.1 Autotrophs convert carbon dioxide into carbohydrates and other carbon compounds
The element carbon is the cornerstone of life as we know it. Carbon is such a crucial element of living organisms that it is part of the definition of living thing. Not only carbon is found in the biosphere in organic molecules such as carbohydrates, protein and lipids, it is also found in the atmosphere and in the lithosphere as fossil fuels in rocks. The biosphere refers to all places where life is found, and the lithosphere refers to all places where rocks are found.
Petroleum is rich in carbon because it originated from partially decomposed organism that died year ago.
Carbon is constantly being cycled between living organisms and inorganic processes that allow the carbon to be available.
Ex: Carbon atoms make up a giraffe cells, she eats organic materials and when cell respiration is complete carbon dioxide is released into the atmosphere and when giraffes dies, its body will be eaten by scavengers. Some of the carbon of the giraffe will go back to the atmosphere.
The element carbon is the cornerstone of life as we know it. Carbon is such a crucial element of living organisms that it is part of the definition of living thing. Not only carbon is found in the biosphere in organic molecules such as carbohydrates, protein and lipids, it is also found in the atmosphere and in the lithosphere as fossil fuels in rocks. The biosphere refers to all places where life is found, and the lithosphere refers to all places where rocks are found.
Petroleum is rich in carbon because it originated from partially decomposed organism that died year ago.
Carbon is constantly being cycled between living organisms and inorganic processes that allow the carbon to be available.
Ex: Carbon atoms make up a giraffe cells, she eats organic materials and when cell respiration is complete carbon dioxide is released into the atmosphere and when giraffes dies, its body will be eaten by scavengers. Some of the carbon of the giraffe will go back to the atmosphere.
carbon dioxide in solution
4.3.2 In aquatic ecosystems, carbon is present as dissolved carbon dioxide and hydrogen carbonate ions
Carbon dioxide is soluble in water. It can either remain in water as a dissolved gas or it can combine with water to form carbonic acid. Carbonic acid can dissociate to form hydrogen and hydrogen carbonate ions. This explains how carbon dioxide can reduce the pH of water.
Both dissolved carbon dioxide and hydrogen carbonate ions are absorbed by aquatic plants and other autotrophs that live in water. They use them to make carbohydrates and other carbon compounds.
Carbon dioxide is soluble in water. It can either remain in water as a dissolved gas or it can combine with water to form carbonic acid. Carbonic acid can dissociate to form hydrogen and hydrogen carbonate ions. This explains how carbon dioxide can reduce the pH of water.
Both dissolved carbon dioxide and hydrogen carbonate ions are absorbed by aquatic plants and other autotrophs that live in water. They use them to make carbohydrates and other carbon compounds.
absorption of carbon dioxide
4.3.3 Carbon dioxide diffuses from the atmosphere or water into autotrophs
Autotrophs use carbon dioxide in the production of carbon compounds by photosynthesis or other processes. This reduces the concentration of carbon dioxide inside autotrophs and the air or water around. Carbon dioxide therefore diffuses from the atmosphere or water into autotrophs. In plants with leaves this diffusion usually happens through stomata in the underside of the leaves. In aquatic plants the entire surface of the leaves and stems is usually permeable to carbon dioxide, so diffusion can be through any part of these parts of the plant
Autotrophs use carbon dioxide in the production of carbon compounds by photosynthesis or other processes. This reduces the concentration of carbon dioxide inside autotrophs and the air or water around. Carbon dioxide therefore diffuses from the atmosphere or water into autotrophs. In plants with leaves this diffusion usually happens through stomata in the underside of the leaves. In aquatic plants the entire surface of the leaves and stems is usually permeable to carbon dioxide, so diffusion can be through any part of these parts of the plant
release of carbon dioxide from the cell respiration
4.3.4 Carbon dioxide is produced by respiration and diffuses out of organisms into water or the atmosphere
Carbon dioxide is a waste product of aerobic cell respiration. it is produced in all cells that carry aerobic respiration. These can be grouped according to the trophic level of the organism:
Carbon dioxide is a waste product of aerobic cell respiration. it is produced in all cells that carry aerobic respiration. These can be grouped according to the trophic level of the organism:
- Non-photosynthetic cells in producers for example root cells in plants
- animals cells
- Saprotrophs such as fungi that decompose dead organic matter
methanogenesis
4.3.5 Methane is produced from organic matter in anaerobic conditions by methanogenic archaeans and some diffuse into the atmosphere or accumulates in the ground.
Alessandro Volta collected bubbles of gas emerging from mud in a reed bed on margins and found that it was inflammable. There he discovered methane. Which is produced widely in anaerobic environments as it is a waste product of a type of anaerobic respiration.
3 different groups of anaerobic prokaryotes are involved:
1. Bacteria that convert organic matte into a mixture of organic acids, alcohol , CO2 and hydrogen
2. Bacteria that use the organic acids and alcohol to produce acetate , CO2 and Hydrogen.
3. Archaeans that produce methane from CO2, hydrogen and acetate. They do it by two chemical reactions:
Alessandro Volta collected bubbles of gas emerging from mud in a reed bed on margins and found that it was inflammable. There he discovered methane. Which is produced widely in anaerobic environments as it is a waste product of a type of anaerobic respiration.
3 different groups of anaerobic prokaryotes are involved:
1. Bacteria that convert organic matte into a mixture of organic acids, alcohol , CO2 and hydrogen
2. Bacteria that use the organic acids and alcohol to produce acetate , CO2 and Hydrogen.
3. Archaeans that produce methane from CO2, hydrogen and acetate. They do it by two chemical reactions:
The archaeans in this group are methanogenic, they carry out methanogenic in many anaerobic environments:
-Mud along the shores and in the bed f lakes
-Swamps, mires, mangrove forest and other wetlands where the soil or peat deposits are waterlogged.
-Landfill sites where organic matter is in wastes that have been buried.
Methane produced from organic waste in anaerobic digesters is not allowed to escape and instead is burned as a fuel.
-Mud along the shores and in the bed f lakes
-Swamps, mires, mangrove forest and other wetlands where the soil or peat deposits are waterlogged.
-Landfill sites where organic matter is in wastes that have been buried.
Methane produced from organic waste in anaerobic digesters is not allowed to escape and instead is burned as a fuel.
oxidation of methane
Methane is oxidized to carbon dioxide and water in the atmosphere.
Molecules of methane released into the atmosphere persist there for only 12 years because it is naturally oxidized in the stratosphere. Monatomic oxygen and highly reactive hydroxyl radicals are involved in methane oxidation. This explains why in the atmosphere the is no much concentration of methane despite the large amount produced.
Molecules of methane released into the atmosphere persist there for only 12 years because it is naturally oxidized in the stratosphere. Monatomic oxygen and highly reactive hydroxyl radicals are involved in methane oxidation. This explains why in the atmosphere the is no much concentration of methane despite the large amount produced.
peat formation
4.3.6 Peat forms when organic matter is not fully decomposed because of acidic and/or anaerobic conditions in waterlogged soils.
In many soils organic matte such as dead leaves are digested by saprotrophic bacteria and fungi. Saprotrophs obtain the oxygen they need for respiration form air spaces in the soil. In some environments water is unable to drain out of soils so they become waterlogged and anaerobic, Saprotrophs cannot live in these conditions so dead organic matter is not fully decomposed. This is called partial decomposition and large quantities of them have accumulated in some ecosystems and become compressed to form dark brown acidic material called peat, about 3% of the earths land surface is covered by peat, the total quantities of this materials are immense.
In many soils organic matte such as dead leaves are digested by saprotrophic bacteria and fungi. Saprotrophs obtain the oxygen they need for respiration form air spaces in the soil. In some environments water is unable to drain out of soils so they become waterlogged and anaerobic, Saprotrophs cannot live in these conditions so dead organic matter is not fully decomposed. This is called partial decomposition and large quantities of them have accumulated in some ecosystems and become compressed to form dark brown acidic material called peat, about 3% of the earths land surface is covered by peat, the total quantities of this materials are immense.
fossilized organic matter.
4.3.7 Partially decomposed organic matter from past geological eras was converted either into coal or into oil and gas that accumulates in porous rocks.
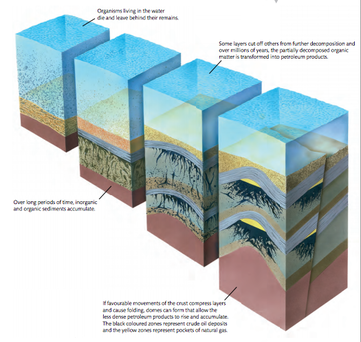
Carbon and some compounds of carbon are chemically very stable and can remain unchanged in rocks for hundreds of millions of years, there are large deposits of carbon from the past geological eras and these deposits are the results of incomplete decomposition of organic matter and its burial in sediments that became rocks.
Carbon and some compounds of carbon are chemically very stable and can remain unchanged in rocks for hundreds of millions of years, there are large deposits of carbon from the past geological eras and these deposits are the results of incomplete decomposition of organic matter and its burial in sediments that became rocks.
- Coal: it is formed when deposits of peat are buried under other sediments, the peat is compressed and heated gradually turning into coal.
- Oil and natural gases: They are formed in the mud at the bottom of the sea and lakes. Conditions are usually anaerobic and so decomposition often is incomplete, as more mud or other sediments are deposits the partially decomposed matter is compressed and heated. Chemical changes occur which produce complex mixture of liquid carbon compounds or gases. We call this mixtures crude oil and natural gas. Methane forms the largest part of natural gas, which can be found in deposits of porous rocks.
combustion
4.3.8 Carbon dioxide is produced by the combustion of biomass and fossilized organic matter
If organic matter is heated to its ignition temperature in the presence of oxygen it will set light and burn. The oxidation reactions that occur are called combustion. The products of complete combustion are carbon dioxide and water. In some parts of the world it is natural to be periodic fires in forest and grasslands (CO2 is produced there). In those parts the organisms are often used to fires and communities regenerates rapidly. In other areas natural fires are very unusual, caused by humans.
Coal, oil and natural gas are different forms of fossilized organic matter, they are all burned into fuels.
If organic matter is heated to its ignition temperature in the presence of oxygen it will set light and burn. The oxidation reactions that occur are called combustion. The products of complete combustion are carbon dioxide and water. In some parts of the world it is natural to be periodic fires in forest and grasslands (CO2 is produced there). In those parts the organisms are often used to fires and communities regenerates rapidly. In other areas natural fires are very unusual, caused by humans.
Coal, oil and natural gas are different forms of fossilized organic matter, they are all burned into fuels.
limestone
4.3.9 Animals such as reef-building corals and Mollusca have hard parts that are composed of calcium carbonate and can become fossilized in limestone.
Some animals have hard body parts composed of calcium carbonate (CaCO3)
In shallowed areas calcium carbonate is also deposited by precipitation in the water, the results are limestone rocks where the deposited hard parts of animals are often visible as fossils.
Some animals have hard body parts composed of calcium carbonate (CaCO3)
- Mollusc shells contain calcium carbonate
- Hard corals
In shallowed areas calcium carbonate is also deposited by precipitation in the water, the results are limestone rocks where the deposited hard parts of animals are often visible as fossils.
carbon cycle diagram
Theory of knowledge: The precautionary principle is meant to guide decision-making in conditions where a lack of certainty exits. Is certainty ever possible in the natural science?
No, scientific claims are based largely on inductive observations, absolute certainty is almost impossible to establish.This is the reason why in labs we have to include the uncertainties in the tables, because we as humans cannot be 100% exact in the results. There is always at least the most minimum error. Absolute truth is just like this, there is no absolute truth however there are things that are really close to it, they are not, just like in science even though a result might be really precise, we cannot be absolutely certain of it.