cell respiration hl
All the information is given by the power points provided in class and Pearson's book
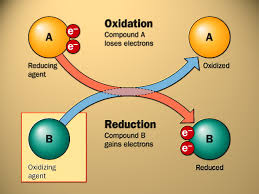
8.1.1 Cell respiration involves the oxidation and reduction of electron carriers.
OXIDATION: loss of electrons but gain oxygen
REDUCTION: gain of electrons but losses oxygen
ELECTRON CARRIERS: NAD (NADH+) OR FAD (FADH+)
8.1.7 Energy released by oxidation reactions is carried to the cristae of the mitochondria by reduced NAD and FAD.
8.1.1 Cell respiration involves the oxidation and reduction of electron carriers.
OXIDATION: loss of electrons but gain oxygen
REDUCTION: gain of electrons but losses oxygen
ELECTRON CARRIERS: NAD (NADH+) OR FAD (FADH+)
8.1.7 Energy released by oxidation reactions is carried to the cristae of the mitochondria by reduced NAD and FAD.
aerobic respiration has 5 processes
1. GLYCOLYSIS
8.1.2 Phosphorylation of molecules makes them less stable.
8.1.3 In glycolysis, glucose is converted to pyruvate in the cytoplasm.
Two molecules of pyruvate of C3 each one.
8.1.4 Glycolysis gives a small net gain of ATP without the use of oxygen.
In Glycolysis 2ATP is used to break the glucose in 2, but then it gains 4 ATP. 2 per pyruvate
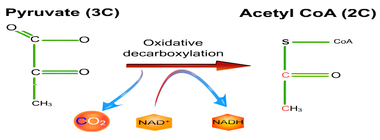
2. LINK REACTION
8.1.5 In aerobic cell respiration, pyruvate is decarboxylated and oxidized, and converted into acetyl compound and attached to
coenzyme A to form acetyl coenzyme A in the link reaction.
In link reaction:
•Oxidative decarboxylation of pyruvate
•Oxidative: NAD + -> NADH + H+
•Decarboxylation: pyruvate loses 1 CO2
•Forming a 2-carbon acetyl fragment, carried by CoA as acetyl-coenzyme (acetyl CoA)
8.1.2 Phosphorylation of molecules makes them less stable.
8.1.3 In glycolysis, glucose is converted to pyruvate in the cytoplasm.
Two molecules of pyruvate of C3 each one.
8.1.4 Glycolysis gives a small net gain of ATP without the use of oxygen.
In Glycolysis 2ATP is used to break the glucose in 2, but then it gains 4 ATP. 2 per pyruvate
2. LINK REACTION
8.1.5 In aerobic cell respiration, pyruvate is decarboxylated and oxidized, and converted into acetyl compound and attached to
coenzyme A to form acetyl coenzyme A in the link reaction.
In link reaction:
•Oxidative decarboxylation of pyruvate
•Oxidative: NAD + -> NADH + H+
•Decarboxylation: pyruvate loses 1 CO2
•Forming a 2-carbon acetyl fragment, carried by CoA as acetyl-coenzyme (acetyl CoA)
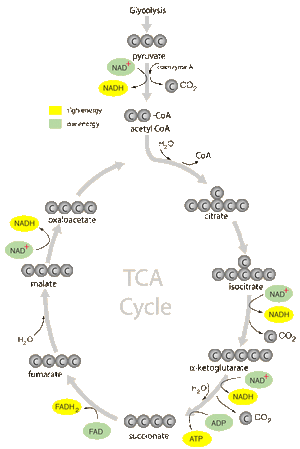
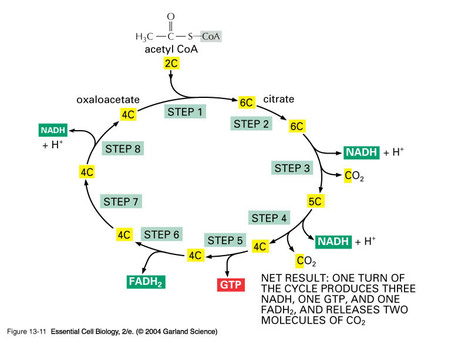
3. KREBS CYCLE (TCA)
8.1.6 In the Krebs cycle, the oxidation of acetyl group is coupled to the reduction of hydrogen carriers, liberating carbon dioxide.
•Matrix of mitochondrion
•2-carbon acetyl joins with 4-carbon molecule, forming 6-carbon molecule
•Oxidized and releases 2 CO2
•PRODUCTS OF THE KREBS CYCLE: Reduction of ADP to ATP, FAD to FADH2, 3 NAD to 3 NADH + H+
4. ELECTRON TRANSPORT CHAIN
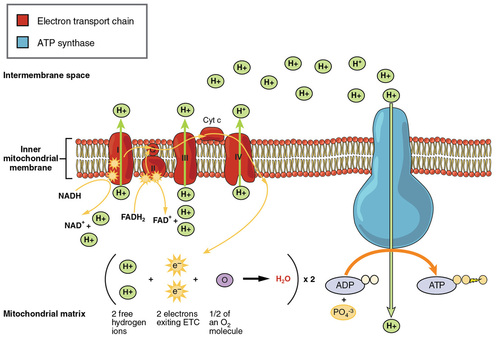
8.1.8 Transfer of electrons between carriers in the electron transport chain in the membrane of the cristae is coupled to proton pumping.
8.1.10 Oxygen is needed to bind with the free protons to maintain the hydrogen gradient, resulting in the formation of water.
8.1.11 The structure of the mitochondrion is adapated to the function it performs.
•NADH + H+ arrives at the first carrier and transfers 2 e-s and 1 H+, another H+ is picked up from the matrix
•the 2 e-s and 2 H+s are carried from the matrix to the inter-membrane space and 2H+s are carried into the inter-membrane space
•the 2 e-s return, pick up another pair of H+s and repeat the trip 2 more times, for a total of 3 round trips
•FADH2 enters into the ETS alter, and pumps only 2 pairs of H+s into the inter-membrane space
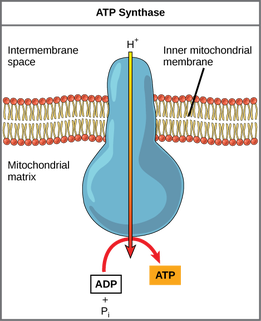
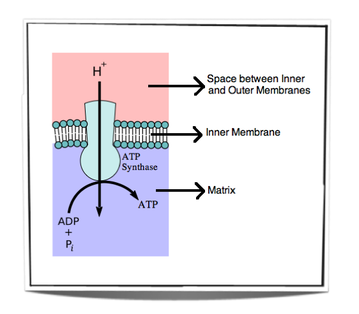
8.1.9 In chemiosmosis, protons diffuse through ATP synthase to generate ATP.
•Oxygen is the final electron acceptor (has high electronegativity)
•accepts 2e- and 2H+ ->2H atom
•Forms 2H + O -> H2O
8.1.8 Transfer of electrons between carriers in the electron transport chain in the membrane of the cristae is coupled to proton pumping.
8.1.10 Oxygen is needed to bind with the free protons to maintain the hydrogen gradient, resulting in the formation of water.
8.1.11 The structure of the mitochondrion is adapated to the function it performs.
•NADH + H+ arrives at the first carrier and transfers 2 e-s and 1 H+, another H+ is picked up from the matrix
•the 2 e-s and 2 H+s are carried from the matrix to the inter-membrane space and 2H+s are carried into the inter-membrane space
•the 2 e-s return, pick up another pair of H+s and repeat the trip 2 more times, for a total of 3 round trips
•FADH2 enters into the ETS alter, and pumps only 2 pairs of H+s into the inter-membrane space
8.1.9 In chemiosmosis, protons diffuse through ATP synthase to generate ATP.
•Oxygen is the final electron acceptor (has high electronegativity)
•accepts 2e- and 2H+ ->2H atom
•Forms 2H + O -> H2O
5.
Oxidative
Phosphorylation
•ATP phosphorylation:
ADP + P (phosphate) -> ATP
•Chemiosmosis : coupling of ATP synthesis to electron transport
•H+ movement across the membrane causes release of energy, which is used by ATP synthase to create ATP
•Therefore: H+: remains in the intermembrane space, lowering pH and contributing to the chemiosmotic gradient --> ATP phosphorylation
•H+ diffuse down chemiosmotic gradient from intermembrane space (pH = 4) through proton channel and into matrix (pH = 8)
•Product
–each NADH + H+ pumped 3 pairs of H+s, which produces 3 ATP
–each FADH2 pumps 2 pairs of H+s, which produces 2 ATP
•ATP phosphorylation:
ADP + P (phosphate) -> ATP
•Chemiosmosis : coupling of ATP synthesis to electron transport
•H+ movement across the membrane causes release of energy, which is used by ATP synthase to create ATP
•Therefore: H+: remains in the intermembrane space, lowering pH and contributing to the chemiosmotic gradient --> ATP phosphorylation
•H+ diffuse down chemiosmotic gradient from intermembrane space (pH = 4) through proton channel and into matrix (pH = 8)
•Product
–each NADH + H+ pumped 3 pairs of H+s, which produces 3 ATP
–each FADH2 pumps 2 pairs of H+s, which produces 2 ATP
video full explanation:
Guidance: The names of the intermediate compounds in glycolysis and the Krebs cycle are required.
Theory of knowledge: Peter Mitchell’s chemiosmotic theory encountered years of opposition before it was finally accepted. For what reasons does falsification not always result in an immediate acceptance of new theories or a paradigm shift?
Falsification is a process in which scientist experiment and test the hypothesis, its results can led to falsificate a theory or to support it. Sometimes this process has to be repeated several times until it is accepted by scientist.
"Kuhn said that falsifiability had played almost no role in scientific revolutions. He argued that scientists working in a field resist the alternative interpretations of 'outsiders', and tenaciously defend their world view by continually elaborating their shared theory; "normal science often suppresses fundamental novelties because they are necessarily subversive of its basic commitments".
http://en.citizendium.org/wiki/scientific_method
Theory of knowledge: Peter Mitchell’s chemiosmotic theory encountered years of opposition before it was finally accepted. For what reasons does falsification not always result in an immediate acceptance of new theories or a paradigm shift?
Falsification is a process in which scientist experiment and test the hypothesis, its results can led to falsificate a theory or to support it. Sometimes this process has to be repeated several times until it is accepted by scientist.
"Kuhn said that falsifiability had played almost no role in scientific revolutions. He argued that scientists working in a field resist the alternative interpretations of 'outsiders', and tenaciously defend their world view by continually elaborating their shared theory; "normal science often suppresses fundamental novelties because they are necessarily subversive of its basic commitments".
http://en.citizendium.org/wiki/scientific_method
| 3.7_8.1_cell_respiration.pptx | |
| File Size: | 1960 kb |
| File Type: | pptx |