translation hl
Nature of science: Developments in scientific research follow improvements in computing—the use of computers has enabled scientists to make advances in bioinformatics applications such as locating genes within genomes and identifying conserved sequences. (3.7)
Understandings
• Initiation of translation involves assembly of the components that carry out the process.
• Synthesis of the polypeptide involves a repeated cycle of events.
• Disassembly of the components follows termination of translation.
• Free ribosomes synthesize proteins for use primarily within the cell.
• Bound ribosomes synthesize proteins primarily for secretion or for use in lysosomes.
• Translation can occur immediately after transcription in prokaryotes due to the absence of a nuclear membrane.
• The sequence and number of amino acids in the polypeptide is the primary structure.
• The secondary structure is the formation of alpha helices and beta pleated sheets stabilized by hydrogen bonding.
• The tertiary structure is the further folding of the polypeptide stabilized by interactions between R groups.
• The quaternary structure exists in proteins with more than one polypeptide chain.
Application and skills:
• Application: tRNA-activating enzymes illustrate enzyme–substrate specificity and the role of phosphorylation.
Skill: The use of molecular visualization software to analyze the structure of eukaryotic ribosomes and a tRNA molecule.
Guidance:
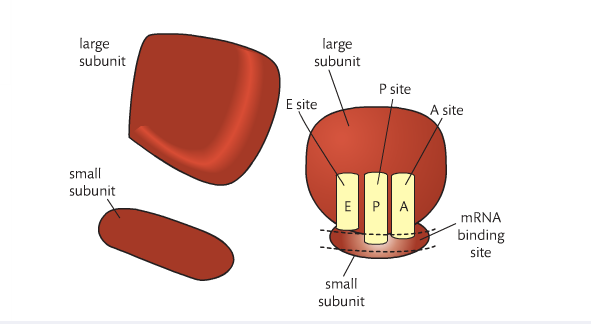
• Names of the tRNA binding sites are expected as well as their roles.
• Examples of start and stop codons are not required.
• Polar and non-polar amino acids are relevant to the bonds formed between R groups.
• Quaternary structure may involve the binding of a prosthetic group to form a conjugated protein.
Understandings
• Initiation of translation involves assembly of the components that carry out the process.
• Synthesis of the polypeptide involves a repeated cycle of events.
• Disassembly of the components follows termination of translation.
• Free ribosomes synthesize proteins for use primarily within the cell.
• Bound ribosomes synthesize proteins primarily for secretion or for use in lysosomes.
• Translation can occur immediately after transcription in prokaryotes due to the absence of a nuclear membrane.
• The sequence and number of amino acids in the polypeptide is the primary structure.
• The secondary structure is the formation of alpha helices and beta pleated sheets stabilized by hydrogen bonding.
• The tertiary structure is the further folding of the polypeptide stabilized by interactions between R groups.
• The quaternary structure exists in proteins with more than one polypeptide chain.
Application and skills:
• Application: tRNA-activating enzymes illustrate enzyme–substrate specificity and the role of phosphorylation.
Skill: The use of molecular visualization software to analyze the structure of eukaryotic ribosomes and a tRNA molecule.
Guidance:
• Names of the tRNA binding sites are expected as well as their roles.
• Examples of start and stop codons are not required.
• Polar and non-polar amino acids are relevant to the bonds formed between R groups.
• Quaternary structure may involve the binding of a prosthetic group to form a conjugated protein.
initiation of translation
Initiation of translation involves assembly of the components that carry out the process.
To begin the process of translation, and mRNA molecule binds to the small ribosomal subunit at an mRNA binding site. An initiator tRNA carrying methionine binds at the start of the condon AUG
The large ribosomal subunit then binds to the small one
The initiator tRNA is in the Psite, the next codon signals another tRNA to bind. It occupies the A site. A peptide bond is formed between the 2 amino acids in the P and A site
To begin the process of translation, and mRNA molecule binds to the small ribosomal subunit at an mRNA binding site. An initiator tRNA carrying methionine binds at the start of the condon AUG
The large ribosomal subunit then binds to the small one
The initiator tRNA is in the Psite, the next codon signals another tRNA to bind. It occupies the A site. A peptide bond is formed between the 2 amino acids in the P and A site
elongation of the polypeptide
• Synthesis of the polypeptide involves a repeated cycle of events.
Following the initiation, elongation occurs through a series of repeated steps. The ribosome translocates 3 bases along the mRNA. moving the tRNA in the p site to the E site, freeing it and allowing a tRNA with the appropriate anticodon to bind to the next codon and occupy the vacant A site
termination of translation
• Disassembly of the components follows termination of translation.
The process continues until a stop codon is reached when the free polypeptide is released. Note that he direction of the movement along the mRNA is from 5' to 3'.
free ribosomes
• Free ribosomes synthesize proteins for use primarily within the cell.
In eukaryotes proteins function in a particular cellular compartment. Proteins are synthesized either in the cytoplasm or at the endoplasmic reticulum depending on the final destination of the protein. Translation occurs more commonly in the cytosol. Proteins destined for the use in the cytoplasm, mitochondria and chloroplast are synthesized by ribosomes free in the cytoplasm
bound ribosomes
• Bound ribosomes synthesize proteins primarily for secretion or for use in lysosomes.
In eukaryotic cells, thousand of proteins are made. In many cases proteins perform a function within a specific compartment of the cell or they are secreted. Proteins must therefore be sorted so that they end up in the correct location. Proteins that are destined for use in the ER, the golgi apparatus, lysosomes and plasma membrane or an outside the cell are synthesized by ribosomes bound to the ER.
Whether the ribosome is free in the cytosol or bound to the ER depends on the presence of a signal sequence on the polypeptide being translated. AS the signal sequence is creates it becomes bound to a signal recognition protein that stops the translation until it can bind to a receptor on the surface of the ER. Once this happen, translation begins again with the polypeptide moving into the lumen of the ER as it is created.
the coupling of transcription and translation prokaryotes
• Translation can occur immediately after transcription in prokaryotes due to the absence of a nuclear membrane.
In eukaryotes, cellular fucntions are compartmented whereas in prokaryotes htey are not. Once transcription is complete in eukaryotes, the transcript is modified in several ways before exiting the nucleus. Thus there is a delay between transcription and translation due to compartmentalization. In prokaryotes, as soon as the mRNA is transcribed the translation begins.
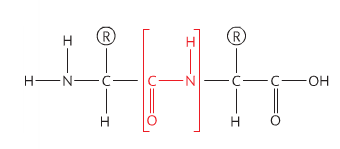
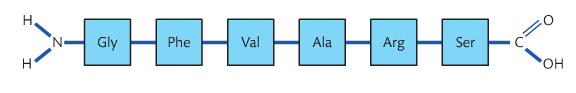
primary structure
• The sequence and number of amino acids in the polypeptide is the primary structure.
A chain of amino acids is called a polypeptide. Given that 20 aminoacids can be combined in any sequence, it should not be surprising that there is a huge diversity of proteins. The sequence of aminoacids in a polypeptide is termed its primary structure.
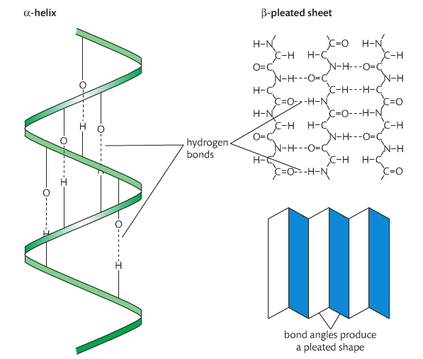
secondary structure
• The secondary structure is the formation of alpha helices and beta pleated sheets stabilized by hydrogen bonding.
Because the chain of amino acids in a polypeptide has a polar covalent bond within its backbone, it tends to fold in such a way that hydrogen bonds from between the carboxyl group (C-O) of one residue and the aminoacids group (N-H) of another part of the chain. This results in the formation of patterns within the polypeptide called secondary structures. The a helix and the B pleated sheet are examples of secondary structures.
tertiary structure
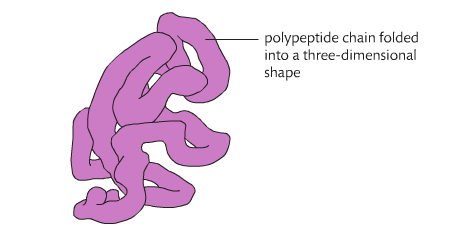
• The tertiary structure is the further folding of the polypeptide stabilized by interactions between R groups.
Tertiary structures refers to the overall 3 dimensional shape of the protein. This shape is a consequence of the interaction of R groups with one another and with the surrounding water medium. There are several different interactions:
-Positively charged R groups will interact with negatively charged R groups
-Hydrophobic amino acids will orientate themselves towards the centre of the polypeptide to avoid contact to water while hydrophilic aminoacids will orientate themselves outward.
-Polar R groups will form hydrogen bonds
-The R groups of the amino acids cysteine can form a covalent bond with the R group of another cysteine, forming a disulphide bridge
quaternary structure
• The quaternary structure exists in proteins with more than one polypeptide chain.
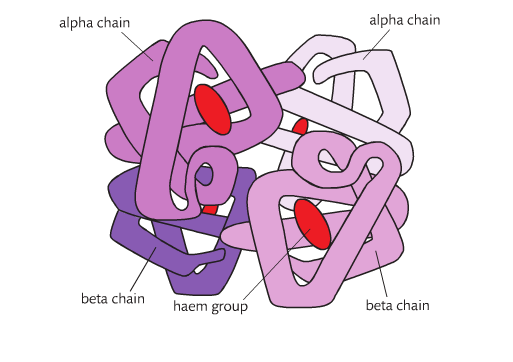
Proteins can be formed form a single polypeptide chain or from more than one polypeptide chain. Lysozyme is composed of a single chains so lysozyme is both a polypeptide and a protein. Insulin is formed out of 2 polypeptides.Hemoglobin is made up 4 chains.
Quaternary structures refers to the way polypeptides fit together when there is more than one chain. It also refers to the addition of a non-polypeptide components. The 4 structure of a hemoglobin consists of 4 polypeptides chains and 4 heme groups
The biological activity of a protein is related to its primary, secondary, tertiary and quaternary structure. Certain treatments such as exposure to high temps or changes to the pH can cause alterations of the structures of a protein and therefore its biological activity.