NITROGEN AND PHOSPHORUS CYCLES
|
Video explanation:
|
|
Source: Chapter C Ecology notes, Oxford text.
C6.1 Nitrogen fixing bacteria convert atmospheric nitrogen to ammonia
C6.2 Rhizobium associates with roots in a mutualistic relationship
C6.3 In the absence of oxygen denitrifying bacteria reduce nitrate in the soil
C6.4 Phosphorus can be added to the phosphorus cycle by application of fertilizer or removed by the harvesting of agricultural crops.
C6. 5 The rate of turnover in the phosphorus cycle is much lower than the nitrogen cycle
C6.6 Availability of phosphate may become limiting to agriculture in the future.
C6.7 Leaching of minerals nutrients from agricultural land into rivers causes eutrophication and leads to increased biochemical oxygen demands
Applications
-The impact of waterlogging on the nitrogen cycle
-Insectivorous plants as an adaptation for low nitrogen availability in waterlogged soils
Skills
-Drawing and labelling a diagram of the nitrogen cycle
-Asses the nutrients content of a soil sample
Nature of science
-Assessing risks and benefits of scientific research: agricultural practices can disrupt the phosphorus cycle.
C6.1 Nitrogen fixing bacteria convert atmospheric nitrogen to ammonia
C6.2 Rhizobium associates with roots in a mutualistic relationship
C6.3 In the absence of oxygen denitrifying bacteria reduce nitrate in the soil
C6.4 Phosphorus can be added to the phosphorus cycle by application of fertilizer or removed by the harvesting of agricultural crops.
C6. 5 The rate of turnover in the phosphorus cycle is much lower than the nitrogen cycle
C6.6 Availability of phosphate may become limiting to agriculture in the future.
C6.7 Leaching of minerals nutrients from agricultural land into rivers causes eutrophication and leads to increased biochemical oxygen demands
Applications
-The impact of waterlogging on the nitrogen cycle
-Insectivorous plants as an adaptation for low nitrogen availability in waterlogged soils
Skills
-Drawing and labelling a diagram of the nitrogen cycle
-Asses the nutrients content of a soil sample
Nature of science
-Assessing risks and benefits of scientific research: agricultural practices can disrupt the phosphorus cycle.
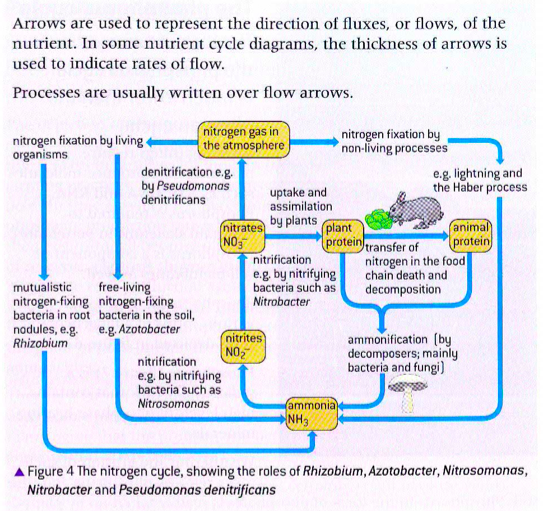
nitrogen cycle
nitrogen fixation
The atmosphere is 78% nitrogen gas in the form of diatomic molecule N2. Nitrogen in this form cannot be taken up by plants. Nitrogen would quickly become a limiting factor for ecosystems if it were not for the bacteria involved in the nitrogen cycle. The bacteria Rhizobium and Azotobacter can FIX nitrogen gas and convert it to ammonia (NH3), a form that living things can use. Once in this form, it can be absorbed by plants and then it can enter food chains.
nitrogen fixation by rhizobium
The bacteria of the genus Rhizobium convert atmospheric nitrogen into a usable organic form of nitrogen. These bacteria are often not free-living but live in a close symbiotic association in the roots of plants such as the legume family (soil beans). Here both organisms benefit, the symbiosis is an example of mutualism.
The bacteria converts (by Fixation) atmospheric nitrogen in the soil to ammonia (NH3). The host plant cannot carry out this process itself, but it is vital fro the production of amino acids, the building blocks of proteins.
Rhizobium infect the plant through root hairs, forming an infection thread which conveys them from the entry point to the nodule site. Once they are within the nodule, they divide repeatedly and swell.
The bacteria converts (by Fixation) atmospheric nitrogen in the soil to ammonia (NH3). The host plant cannot carry out this process itself, but it is vital fro the production of amino acids, the building blocks of proteins.
Rhizobium infect the plant through root hairs, forming an infection thread which conveys them from the entry point to the nodule site. Once they are within the nodule, they divide repeatedly and swell.
denitrification
The ammonia produced by nitrogen fixation is converted into nitrite (NO2) by the genus of bacteria known as Nitrosomonas. These bacteria have a double membrane and use electrons gained form the oxidation of ammonia to produce energy. Nitrites are produced from this oxidation. The energy is used to fix carbon dioxide into organic carbon molecules. This means that nitrosomonas are a genus of chemoautotrophs as they use the energy found in ammonia, which is an inorganic molecule.
Nitrites are converted to nitrates by the genus of bacteria known as nitrobacter (picture bellow) . Nitrobacter sp are an example of chemoautrotrophs as they derive energy from nitrites which are inorganic compounds. They oxidize nitrites as an energy source for carbon fixation and convert it to nitrates. Nitrate is a form of nitrogen that is bioavailable to plants.
Denitrifiction is the reduction of nitrate (NO3) to nitrogen (N2). Denitrifying bacteria such as Pseudomonas denitrificans can use oxygen as an electron acceptor. However when oxygen is in short supply, instead of using O2 as an electron transport, denitrifying bacteria use nitrate as an electron acceptor, releasing gaseous nitrogen as the product. When that occurs the bioavailability of nitrogen in the ecosystem is reduced.
Nitrites are converted to nitrates by the genus of bacteria known as nitrobacter (picture bellow) . Nitrobacter sp are an example of chemoautrotrophs as they derive energy from nitrites which are inorganic compounds. They oxidize nitrites as an energy source for carbon fixation and convert it to nitrates. Nitrate is a form of nitrogen that is bioavailable to plants.
Denitrifiction is the reduction of nitrate (NO3) to nitrogen (N2). Denitrifying bacteria such as Pseudomonas denitrificans can use oxygen as an electron acceptor. However when oxygen is in short supply, instead of using O2 as an electron transport, denitrifying bacteria use nitrate as an electron acceptor, releasing gaseous nitrogen as the product. When that occurs the bioavailability of nitrogen in the ecosystem is reduced.
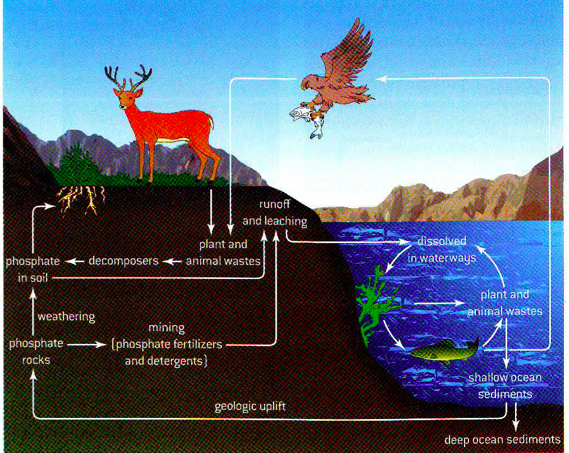
the phosphorus cycle
All living things require phosphorus to produce molecules such as ATP (the P means phosphate), DNA and RNA. Phosphorus is required to maintain skeletons in vertebrates. Phosphorus is a component of cell membranes as well (phospholipid bilayer)
Phosphorite is a sedimentary rock that contains high level of phosphate-bearing minerals
Weathering and erosion of such rocks releases phosphates into the soil. Phosphorus in the form of phosphates is readily taken up by plants, where it enters food chains.
The largest stocks of phosphate occur in marine sediments and in mineral deposits.
Turnover rate refers to the amount of phosphorus released from one stock to another per unit time. Phosphate is only slowly released to ecosystems by weathering and so has a relatively low turnover rate in comparison to nitrogen.
Phosphorite is a sedimentary rock that contains high level of phosphate-bearing minerals
Weathering and erosion of such rocks releases phosphates into the soil. Phosphorus in the form of phosphates is readily taken up by plants, where it enters food chains.
The largest stocks of phosphate occur in marine sediments and in mineral deposits.
Turnover rate refers to the amount of phosphorus released from one stock to another per unit time. Phosphate is only slowly released to ecosystems by weathering and so has a relatively low turnover rate in comparison to nitrogen.
effect of agriculture in soil phosphorus
Human activity impacts the phosphorus cycle. Phosphate is mined and converted to phosphate-based fertilizer. The fertilizer is then transported great distances and applied to crops. Phosphorus in the biomass of corps is transferred from the fields in one are to markets in other areas.
Waterlogging of soils in poorly drained irrigated crops can dissolve phosphate and bring it into solution. Runoff containing phosphate from fertilizer can contribute to fresh waster eutrophication.
Waterlogging of soils in poorly drained irrigated crops can dissolve phosphate and bring it into solution. Runoff containing phosphate from fertilizer can contribute to fresh waster eutrophication.
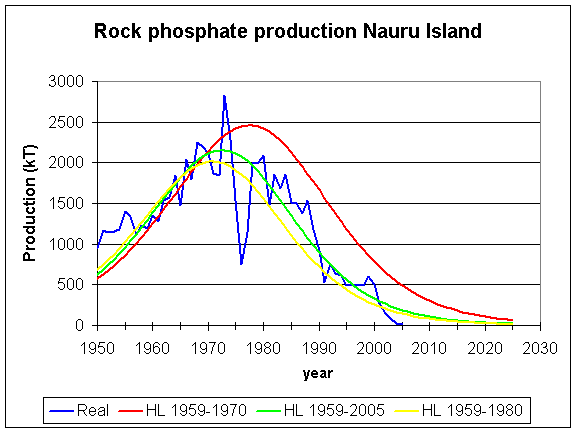
peak phosphorus
The depletion of phosphate resources that can be mined is a concern because of the role it plays in fertilizer fro modern intensive farming practices. Peak phosphorus is the point in time at which the maximum global phosphate production is reached and then begins to fall because of the depletion of reserves. Figure 8 shows a graph of the world phosphate. This graph suggests that the point of peak phosphorus production is approaching.
There is a lack of agreement on the amount of available phosphate rock reserves though many agree that the problem will become acute within 50 to 100 years.
Without fertilizers, famine would most certainly result because yields per unit of farmland would plummet without the addition of fertilizer. There are no alternative sources of phosphate and no synthetic way of creating it, unlike ammonia which can be created by the industrial conversion of plentiful supplies of atmospheric nitrogen.
There is a lack of agreement on the amount of available phosphate rock reserves though many agree that the problem will become acute within 50 to 100 years.
Without fertilizers, famine would most certainly result because yields per unit of farmland would plummet without the addition of fertilizer. There are no alternative sources of phosphate and no synthetic way of creating it, unlike ammonia which can be created by the industrial conversion of plentiful supplies of atmospheric nitrogen.
eutrophication and biochemical oxygen demand
When rain falls on agricultural land, water-soluble nutrients that have been added to crops such as phosphates and nitrates can dissolve in the water and the resulting runoff can enter water courses and streams. In addition to crops, nutrients form manure and urine of livestock can also contribute to nutrient enrichment of bodies of water.
The nutrient enrichment of water is known as eutrophication. The nutrients favour the growth of algae leading to algal blooms (or Lemna) which block the light to the plants bellow.
When the mats of algae die and the plants below them die, it leads to a loss of oxygen because of bacterial activity on the dead organic matter. This is called Biological oxygen demand (BOD). The higher the BOD the more anoxic a body of water becomes limiting the life of fishes under water.
The nutrient enrichment of water is known as eutrophication. The nutrients favour the growth of algae leading to algal blooms (or Lemna) which block the light to the plants bellow.
When the mats of algae die and the plants below them die, it leads to a loss of oxygen because of bacterial activity on the dead organic matter. This is called Biological oxygen demand (BOD). The higher the BOD the more anoxic a body of water becomes limiting the life of fishes under water.