WATER
All the information is from power points provided in class and Pearson's and the study guide books.
KEY POINTS
2.2.1 Water molecules are polar and hydrogen bonds form between them.
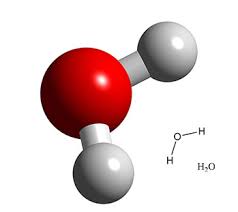
notes: water molecules consist in two hydrogen atoms bonded to an oxygen atom. hydrogen have a positive charge and oxygen negative, therefore they have two poles one + and one -.
A hydrogen bond can form between the positive pole and the negative one, that is called hydrogen bond
2.2.2 Hydrogen bonding and dipolarity explain the cohesive, adhesive, thermal and solvent properties of water.
notes: (explained later)
2.2.3 Substances can be hydrophilic or hydrophobic.
notes: can be hydrophilic if they are in the + pole and love water
can be hydrophobic if they are in the - pole and hate water
KEY POINTS
2.2.1 Water molecules are polar and hydrogen bonds form between them.
notes: water molecules consist in two hydrogen atoms bonded to an oxygen atom. hydrogen have a positive charge and oxygen negative, therefore they have two poles one + and one -.
A hydrogen bond can form between the positive pole and the negative one, that is called hydrogen bond
2.2.2 Hydrogen bonding and dipolarity explain the cohesive, adhesive, thermal and solvent properties of water.
notes: (explained later)
2.2.3 Substances can be hydrophilic or hydrophobic.
notes: can be hydrophilic if they are in the + pole and love water
can be hydrophobic if they are in the - pole and hate water
water chemical structure
hydrogen bonding water
A bond can be form between the positive pole and the negative water pole. That is called " hydrogen bond" .
In liquid water many of these bonds form, giving water properties that make it a very useful substance for living organisms.
In liquid water many of these bonds form, giving water properties that make it a very useful substance for living organisms.
properties of water
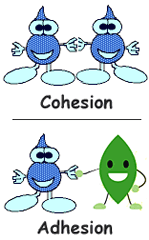
1. COHESION
Water molecules stick to each other because of the hydrogen bonds form between them. ex: to suck columns of water up to the tops of the tallest trees.
2. SOLVENT PROPERTIES
Many different substances dissolve in water because particles with positive or negative charges dissolve, for example sodium ions. Organic substances with polar molecules dissolve, for example glucose . ex: most chemical reactions in living organism take place with all of the substance involved in the reactions dissolved in water. It is also used as a transport medium
3. THERMAL PROPERTIES: HEAT CAPACITY
Water has a large heat capacity, large amount of energy needed to raise its temperature. The energy is needed to break some of the hydrogen bonds.ex: blood which is composed of water carry heat from warmer parts to cooler parts of the body
4.THERMAL PROPERTIES: BOILING POINT
The boiling point of the water is 100C, which is high because to change it from a liquid to a gas all of the hydrogen bonds between water molecules have to be broken. ex: it is almost everywhere on earth
5. THERMAL PROPERTIES: THE COOLING EFFECT OF EVAPORATION
Water can evaporate at temperature below the boiling point. Hydrogen bonds have to be broken to do this. The heat energy needed to break the bonds is taken from the liquid to cooling it down. ex: evaporation of water from plants leaves
Water molecules stick to each other because of the hydrogen bonds form between them. ex: to suck columns of water up to the tops of the tallest trees.
2. SOLVENT PROPERTIES
Many different substances dissolve in water because particles with positive or negative charges dissolve, for example sodium ions. Organic substances with polar molecules dissolve, for example glucose . ex: most chemical reactions in living organism take place with all of the substance involved in the reactions dissolved in water. It is also used as a transport medium
3. THERMAL PROPERTIES: HEAT CAPACITY
Water has a large heat capacity, large amount of energy needed to raise its temperature. The energy is needed to break some of the hydrogen bonds.ex: blood which is composed of water carry heat from warmer parts to cooler parts of the body
4.THERMAL PROPERTIES: BOILING POINT
The boiling point of the water is 100C, which is high because to change it from a liquid to a gas all of the hydrogen bonds between water molecules have to be broken. ex: it is almost everywhere on earth
5. THERMAL PROPERTIES: THE COOLING EFFECT OF EVAPORATION
Water can evaporate at temperature below the boiling point. Hydrogen bonds have to be broken to do this. The heat energy needed to break the bonds is taken from the liquid to cooling it down. ex: evaporation of water from plants leaves
Guidance: Students should know at least one example of a benefit to living organisms of each property of water.
- Water is used as a transport medium
-Water is the medium for metabolic reactions
-Water is used as a transport medium for heat.
-Water can be used as a coolant.
Transparency of water and maximum density at 4C do not need to be included. Comparison of the thermal properties of water and methane assists in the understanding of the significance of hydrogen bonding in water.
- Water is used as a transport medium
-Water is the medium for metabolic reactions
-Water is used as a transport medium for heat.
-Water can be used as a coolant.
Transparency of water and maximum density at 4C do not need to be included. Comparison of the thermal properties of water and methane assists in the understanding of the significance of hydrogen bonding in water.
Theory of knowledge: Claims about the “memory of water” have been categorized as pseudoscientific.
What are the criteria that can be used to distinguish scientific claims from pseudoscientific claims?
The examples showed below, are criteria that are used to distinguish the scientific claims from pseudoscientific claims.
Ex: Biorhythms, which like astrology relied uncritically on birth dates, did not meet the criterion of pseudoscience at the time because there were no alternative explanations for the same observations. The use of this criterion has the consequence that a theory can at one time be scientific and at another pseudoscientific.
Ex: We designate a doctrine a ‘pseudoscience’ only when they perceive themselves to be threatened
What are the criteria that can be used to distinguish scientific claims from pseudoscientific claims?
The examples showed below, are criteria that are used to distinguish the scientific claims from pseudoscientific claims.
Ex: Biorhythms, which like astrology relied uncritically on birth dates, did not meet the criterion of pseudoscience at the time because there were no alternative explanations for the same observations. The use of this criterion has the consequence that a theory can at one time be scientific and at another pseudoscientific.
Ex: We designate a doctrine a ‘pseudoscience’ only when they perceive themselves to be threatened